London: The unsightly effects of scarring following surgery, trauma and burns could be reduced significantly by a new technique involving a single, precisely-timed insulin injection.
In a research breakthrough which potentially could benefit millions of people1 annually, the use of insulin2 to reduce levels of scar tissue is being developed by Dr Claire Linge, Group Leader in Cell Biology at the RAFT3 Research Institute, Northwood, Middlesex.
Preliminary clinical trials have produced very promising results and the technique, for which a patent is pending, will be progressing into full scale clinical trials during the coming year.
Dr Linge is taking forward her pioneering research in partnership with NHS Innovations East 4, the regional hub of the NHS National Innovation Centre. Currently, the hub is assisting Dr Linge with the patent application and negotiating with pharmaceutical companies to progress the research into large-scale trials. The patent process is being funded by RAFT.
Inspiration for this pioneering technique came from previous research conducted by Dr Linge into the effects of insulin on different cell types.
Dr Linge says: In the past, I researched insulins ability to change the way certain types of cell behave. Following my move to RAFT, I developed an interest in the scarring process and recognised similarities between the cells of scar tissue and others that I had researched.
I was struck by the possibility that insulin may possess properties which beneficially affect the development of scar tissue.
Detailed investigation of this theory was successful and, with the support of RAFT, Dr Linge developed a treatment technique that was taken into preliminary clinical trials5 at Mount Vernon Hospital, Middlesex.
Dr Linge explains: For an open wound to heal, the skin cells (fibroblasts) that are normally responsible for day-to-day maintenance become activated, producing proteins that fill in the tissue deficit. The function of these activated fibroblasts is absolutely essential for successful healing.
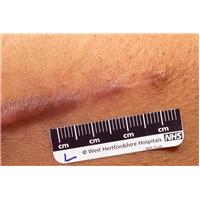
Wounds, such as those caused by surgery or trauma, can often lead to unnecessarily severe scars, with a raised and red appearance. This excess scar tissue is formed by the prolonged presence (for more than a few days) of activated fibroblasts at the wound site.
I have found that a single subcutaneous injection of a low dose of insulin along the margin of a wound restricts the presence of activated fibroblasts to the first few days. This prevents the build up of scar tissue in the first place and thereby results in paler, flatter scars.
At present, there is no acknowledged treatment for scars. Techniques such as pressure bandages are used but their efficacy is not clinically proven.
The pioneering work of Dr Linge was recognised in December 2006 by a panel of eminent biotechnology experts and investment industrialists which awarded her the London Biotechnology Network 2006 BIO-Innovation Award6.
Dr Paul Seabright, Head of Business Development, NHS Innovations East, says: We are delighted to support the work of Dr Linge, which potentially could benefit millions of people and make a major contribution to global healthcare.
Dr Linges work is an inspirational example of the many hundreds of ideas that NHS staff come up with each year to further benefit patient care. As an NHS innovation hub, our role is to help staff develop and implement their innovative technologies and practices in order to improve the quality of service to NHS patients.
Background
1 In the UK alone, during 2004/05, the number of patients who underwent surgical procedures was over six million; 10,000 patients were treated for burns and 56,000 for open wounds caused by trauma.
2 Insulin has been used since the mid-20th century to treat diabetes. More recently, its other properties, including promoting the kind of cell growth that could increase the speed of wound healing, have become more apparent. Dr Linge has shown that a naturally occurring protein will inhibit the development of skin cells responsible for producing scar tissue. Following laboratory and clinical trials, insulin (and its pharmacological mimics) promises to provide a cost-effective, easy-to-use treatment that will reduce the severity of skin trauma, whether caused by accident or surgery, for millions of patients each year.
3 RAFT, The Restoration of Appearance and Function Trust. Registered Research Charity No. 299811. www.raft.ac.uk
4 NHS Innovations East is run by Health Enterprise East Ltd. and is the innovation hub for healthcare in the East of England, part of a national network of NHS innovation hubs and a regional network of innovations hubs, supporting public and private sector healthcare providers. It provides a broad range of professional intellectual property management services to the 40 NHS Trusts across the East of England (Bedfordshire, Cambridgeshire, Essex, Hertfordshire, Norfolk and Suffolk) to enable their employees from all disciplines to identify, evaluate and take forward innovations that can benefit their patients. Health Enterprise East Ltd. is funded by the Department of Health, Department of Trade and Industry and East of England Development Agency.
5 Preliminary clinical trials involved women attending Mount Vernon Hospitals plastic surgery service (at the time run by West Hertfordshire Hospitals NHS Trust, but now managed by the Royal Free Hampstead NHS Trust), for bilateral breast reductions. The operation requires two incisions below each breast. The women who volunteered to participate in the trials were given injections of insulin around the site of one incision while the other incision was not treated, thereby acting as a control. Researchers were able to measure and compare the development of scar tissue on both incisions.
6 Dr Linge received the prestigious London Biotechnology Network’s 2006 BIO-Innovation Award for her pioneering work in using insulin to reduce skin scarring after surgery, trauma or burns. The award was presented by Malcolm Wicks, Minister for Science and Innovation, at the Genesis VI Biotechnology Conference in London. The judges, a panel of eminent biotech and investment industrialists, chose Dr Linge’s entry for its clarity and the progress made by an academic group in getting the technology into a phase 2 clinical trial.